 CDC (2017) recommends that PVAD-shorts be replaced every 72 to 96 hours to prevent infection and phlebitis in adults. They are easy to monitor and can be inserted at the bedside. PVAD-shorts are used for infusions under six days and for solutions that are iso-osmotic or near iso-osmotic (CDC, 2017). The hub of a short intravenous catheter should be attached to IV extension tubing with a needleless cap (Fraser Health Authority, 2014). If a lower extremity is used, remove the peripheral IV and re-site in the upper extremities as soon as possible (CDC, 2017 McCallum & Higgins, 2012). Upper extremities (hands and arms) are the preferred sites for insertion by a specially trained healthcare provider. Dressings serve to keep the site sterile and prevent accidental dislodgement (CDC, 2017). Sites are covered with dressings, which can be a sterile transparent semi-permeable dressing or a gauze dressing if the site is bleeding (RNAO, 2005/2008). 
Principles of asepsis are followed during insertion. Short Peripheral Catheter (SPC)Ī PVAD-short cannula is a common, preferred method for short-term IV therapy in the hospital setting (see Figure 8.1). Peripheral Venous Access Device-Short Cannula (PVAD-Short) a.k.a. Besides observing the site for complications, accessing, flushing, and removal of IVADs, tunneled catheters, and catheters used for hemodialysis require specialized skill and they are not within the scope of this textbook. This section will describe two types of venous access: peripheral IV access and central venous catheters. There are a variety of options available, and a venous access device must be selected based on the duration of IV therapy, type of medication or solution to be infused, and the needs of the patient. In practice it is important to understand the options of appropriate devices available. You can add items from different specialties it just needs to be $125 or more in order to complete the transaction.Safe and reliable venous access for infusions is a critical component of patient care in acute and community health settings. There is a $125 minimum order due to the cost of shipping. The only products we caution customers about are ones that are absorbable or drug coated. The manufacturers sterilize the products after they are packaged and it lasts so many years. Not for human use in the US.Įxpiration dates have mostly to do with sterilization and not the integrity of the product. 
In Perfect sealed condition but past expiration date. Tissue Dilator, 3 ml Applicator Pouch, 2% CHG and 70% IPA ChloraPrep®2 One-Step Solution with Hi-Lite Orange™ Tint,ĥ ml Ampule HCl, 1% Lidocaine Solution, Catheter Clamp Fastener, SharpsAway® Disposal Cup, CSR Wrap,ĭrape: 24 x 36 Inch with 4 Inch Fenestration, Scalpel #11, 2 Gauze Pads: 2 x 2 Inch, 5 Gauze Pads 4 x 4 Inch, Suture: 3-0 Braided Silk with Straight NeedleĪrrow AK-24703 Multi-Lumen Central Venous Catherization Kit Diameter 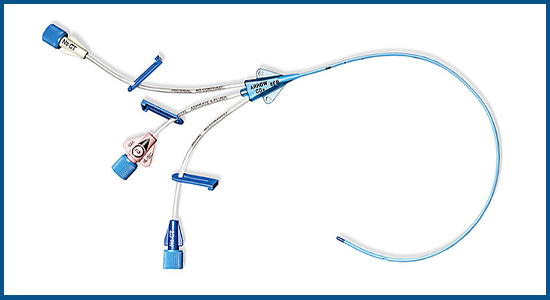
Introducer Needle: 18 G x 2-1/2 Inch XTW and 5 mL Arrow® Raulerson Spring-Wire Introduction Syringe, Injection Needle: 25 G x 1 Inch and 3 mL Luer-Lock Syringe 
Needle, Pressure Transduction Probe, Injection Needle: 22 G x 1-1/2 Inch and 5 mL Luer-Slip Syringe, Part # AK-24703 Exp 2/11 Spring-Wire Guide: 0.032 Inch D X 26-13/16 Inch (Straight Soft Tip on One End – J Tip on Other),Įxtra Thin Wall, Catheter: 18 G X 2-1/2 Inch Radiopaque over 20 G Regular Wall Introducer Arrow AK-24703 Multi-Lumen Central Venous Catherization Kit 7Fr, Triple Lumen 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
